The medical device industry is facing mounting pressure to adopt more rigorous publication management practices as regulatory requirements evolve. The International Society for Medical Publication Professionals (ISMPP) reports how current publication practices, largely shaped for the pharmaceutical industry, often fall short in addressing the unique needs of medical device companies.
While pharmaceutical companies have embraced robust publication planning models, medical device companies have lagged behind, but this is quickly changing as regulatory bodies demand higher standards of data reporting and transparency.
In light of these shifts, medical device companies need to rethink how they approach publication planning and dissemination. One potential solution? Embracing automation to streamline publication efforts and optimize resources.
Let’s dive into the unique publication management challenges that medical device companies face and discover how automation can help.
The unique challenges of medical device publication management
Medical device companies operate in a landscape vastly different from their pharmaceutical counterparts. While the pharmaceutical industry has established a standard approach to publication planning, the medical device sector presents several unique challenges that make this one-size-fits-all approach difficult to implement.
Challenge #1: Diverse medical device product portfolios
Medical devices range from simple surgical tools to highly complex implantable technologies, resulting in a product portfolio that’s far more diverse than that of pharmaceuticals. According to the World Health Organization, there are over 2 million medical devices available globally. This diversity means medical device companies may find it impractical to create publication plans for every product, especially for lower-priority or legacy devices.
How automation helps: Publication automation software can manage multiple publication plans across a diverse range of products, ensuring that even lower-priority devices receive adequate attention. With automation, companies can scale their publication efforts without overwhelming their internal teams.
Challenge #2: Shorter development timelines
Medical devices often have much shorter development timelines compared to pharmaceuticals — typically ranging from 3 to 7 years. This compressed timeline means there is less opportunity to gather and publish key data during development, and the focus tends to shift toward late-stage or post-market publications. Additionally, while pharmaceuticals can demonstrate safety and efficacy with shorter follow-up periods, certain medical devices (e.g., orthopedic implants) require years of post-market data collection to truly prove their long-term safety and effectiveness.
How automation helps: By automating data collection and publication workflows, medical device companies can ensure they are publishing relevant data throughout the device lifecycle — whether it’s pre-market, post-market, or during long-term surveillance. Automation helps keep up with fast-paced development timelines by streamlining the publication process.
Challenge #3: Limited resources and smaller budgets
The medical device industry typically operates with significantly smaller budgets compared to the pharmaceutical sector. This disparity often means medical device companies have smaller, multifunctional teams handling a range of responsibilities — including publication planning, regulatory submissions, and marketing communications.
How automation helps: With smaller teams, medical device companies can benefit from automation to handle repetitive tasks, streamline workflows, and free up resources for more strategic initiatives. Automation reduces the need for large, dedicated publication teams while ensuring consistency and quality across all publications.
How automation improves medical device publication processes
As medical device companies are increasingly expected to adopt more formal publication planning practices, automated publication solutions can provide critical support in overcoming the unique challenges they face.
1. Centralized data management
Automation software allows companies to collect, store, and manage data from multiple sources — preclinical studies, clinical trials, and post-market surveillance — within a single platform. This centralized approach makes it easier to access and use relevant data for various publications, ensuring that nothing is overlooked.
2. Task automation for multi-stakeholder input
Publication planning for medical devices often involves collaboration between different stakeholders. Manually managing these inputs can be time-consuming and error-prone. Task automation ensures that specific responsibilities and deadlines are automatically assigned to the right individuals, preventing bottlenecks and reducing the risk of missed input.
3. Real-time collaboration
Traditional publication processes can suffer from delays as documents are manually passed between reviewers. Automation platforms allow for real-time collaboration, enabling multiple stakeholders to provide input simultaneously without sacrificing version control or creating confusion. Real-time collaboration features speed up the review process and reduce delays, ensuring that publications are completed efficiently and accurately.
4. Audit trails and transparency
Regulatory bodies require transparency in how data is collected, reviewed, and published. This often involves detailed documentation of every step in the publication process, from data gathering to final approval.
Publication management software that offers built-in audit trails can track every action taken during the publication process, from edits to approvals. This ensures complete transparency and reduces the risk of regulatory non-compliance.
Long-term benefits of automating publications
By embracing automation, medical device companies can do more than just meet regulatory requirements — they can also position themselves for long-term success by delivering lasting benefits, such as:
- Maximized resource allocation: Automation helps medical device companies prioritize their publication efforts, allowing them to focus on high-priority products while ensuring that all necessary publications are completed. This optimizes resource allocation, even for smaller teams with limited budgets.
- Consistent publication output: Rather than reacting to the latest clinical trial results, automation helps medical device companies consistently publish data throughout a product’s lifecycle. This ensures a steady flow of information to HCPs and regulatory bodies, helping to build trust and maintain market presence.
- Stronger relationships with HCPs: Healthcare providers rely on published data to make informed decisions about which medical devices to adopt. By using automation to streamline publication efforts, medical device companies can ensure timely, transparent data dissemination that strengthens relationships with HCPs.
The path forward for medical device publications
As the medical device industry adapts to evolving regulatory demands, adopting publication automation software can help streamline processes, reduce inefficiencies, and ensure that all publication activities are aligned with best practices. By embracing automation, medical device companies can overcome many of the challenges they face — maximizing resource efficiency, improving regulatory compliance, and consistently delivering high-quality publications.
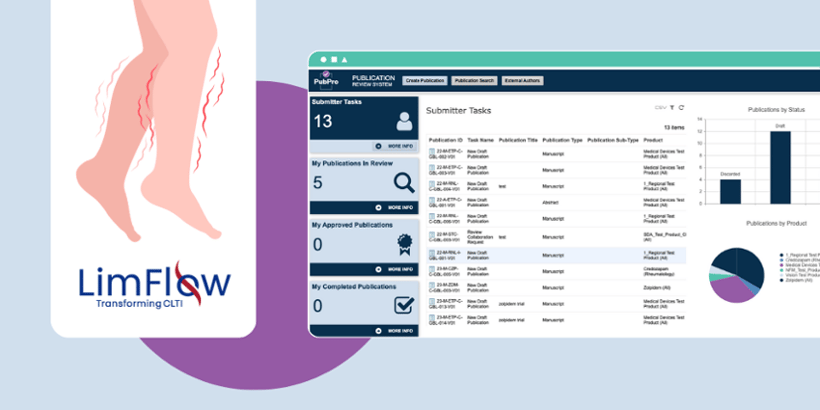
For medical device companies looking to bridge the gap between complex product portfolios and rigorous publication management, automated publication management software, like PubPro, is the key to success.

 Enterprise-level software solutions like iEnvision and PubStrat represent one end of the spectrum.
Enterprise-level software solutions like iEnvision and PubStrat represent one end of the spectrum. On the other end of the spectrum, some teams opt for a piecemeal approach, combining multiple specialized tools for different tasks. This approach might involve using Veeva for document management, separate software for reference management, and manual tools for collaboration.
On the other end of the spectrum, some teams opt for a piecemeal approach, combining multiple specialized tools for different tasks. This approach might involve using Veeva for document management, separate software for reference management, and manual tools for collaboration. Outsourcing medical communications to specialized agencies is another alternative. These agencies bring expertise in scientific writing, regulatory compliance, and effective communication with healthcare professionals and patients.
Outsourcing medical communications to specialized agencies is another alternative. These agencies bring expertise in scientific writing, regulatory compliance, and effective communication with healthcare professionals and patients.
 I’m Alex Reinthal, and as Vice President of Product at BP Logix, I have a unique vantage point on how AI and machine learning technologies will develop in life sciences software.
I’m Alex Reinthal, and as Vice President of Product at BP Logix, I have a unique vantage point on how AI and machine learning technologies will develop in life sciences software.






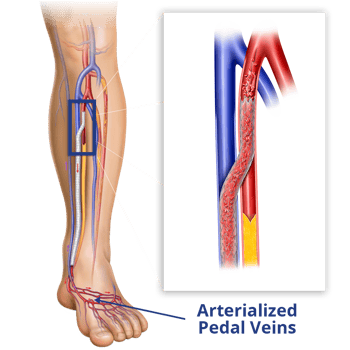 The LimFlow System is a minimally invasive procedure that improves blood flow by creating a bypass around blocked arteries. Our unique approach to treating this debilitating condition offers new hope to those who have been told amputation is their only option.
The LimFlow System is a minimally invasive procedure that improves blood flow by creating a bypass around blocked arteries. Our unique approach to treating this debilitating condition offers new hope to those who have been told amputation is their only option.