Keep IND and NDA submissions on track with Approvia, a document management software designed for regulatory affairs.
Whether it's an Investigational New Drug (IND) or New Drug Application (NDA), managing a document submission process that spans multiple departments can quickly become complicated, increasing the risk of missed deadlines and rejections from the FDA.
Regulatory submissions are not managed together.
Centralize cross-departmental submission processes.
Approvia is a powerful workflow automation solution that centralizes cross-departmental document management, allowing regulatory affairs teams to keep critical FDA submission processes on track.
Approvia groups documents, reviewers, and processes into dossiers and then automatically assigns tasks to users based on their pre-configured role in the process. Role-specific dashboards allow users to see their assigned tasks for active submissions.
Once a dossier is ready to go, send it off to the FDA with Approvia’s electronic submission integration.
Break down information silos and enable seamless communication across teams. Approvia’s collaborative document editor allows submitters and reviewers to work together in real-time and avoid version control issues.
Need to loop in external stakeholders? Deep linking allows external stakeholders to securely access and contribute to documents — no login or password necessary.
Always-on audit tracking logs every action taken during a process, providing unparalleled visibility and accountability.
See every delay, change, and request in one place, and generate reports with a click to prove compliance.
Approvia is designed to be customized to your organization's unique standard operating procedures.
This prevents your users from adopting clunky workarounds or switching between multiple applications.
Our flexible, scalable process automation solutions empower regulatory affairs teams at life science organizations to operate better and do their best possible work.

With Process Director, you get a process engine that works every time.
There's endless possibilities for automating and optimizing other processes, whether you use one of our pre-built use cases or create your own.
Enjoy the greatest bang for your buck as you create more new processes.
Get the convenience of SaaS with the added luxury of a consultative business partnership. We’re there to support you every step of the way.
Get the speed and affordability you need, plus powerful customization options not available in competing regulatory affairs solutions.
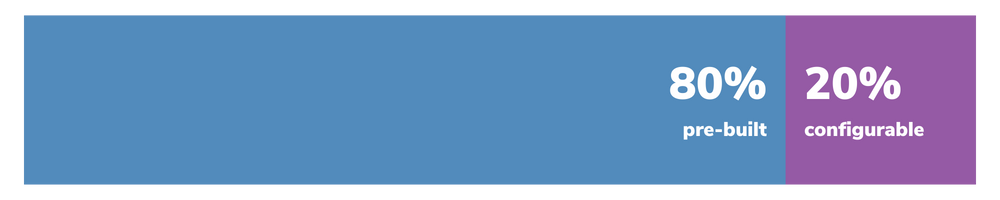
Approvia offers the perfect balance of pre-built functionality and flexibility, with 80% of the solution already built and the remaining 20% configured to your organization's specific requirements.

Approvia groups documents, reviewers, and processes into dossiers and then automatically assigns tasks to users based on their pre-configured role in the process. Role-specific dashboards allow users to see their assigned tasks for active submissions.
Once a dossier is ready to go, send it off to the FDA with Approvia’s electronic submission integration.
Approvia break down information silos and enable seamless communication across teams.
Instead of manually managing edits across multiple stakeholders, Approvia’s collaborative document editor allows submitters and reviewers to work together in real-time and avoid version control issues.
Need to loop in external stakeholders? Deep linking allows external stakeholders to securely access and contribute to documents — no login or password necessary.
Always-on audit tracking logs every action taken during a process, providing unparalleled visibility and accountability. See every delay, change, and request in one place, and generate reports with a click to prove compliance.
Approvia is designed to be customized to your standard operating procedures, which prevents your users from adopting clunky workarounds or switching between multiple applications.